Exceptions management is a critical process that focuses on identifying and resolving data issues that arise during the production and distribution of products. In the pharma industry it directly impacts compliance, quality, operational efficiency, distribution and patient safety.
As trading partners continue to implement and fine-tune their processes to adhere to DSCSA guidelines, the volume of errors related to discrepancies between the data received and the product received in the shipment is expected to increase exponentially. Considering the challenges related to counterfeits, drug shortages and patient safety, efficient handling of exceptions becomes imperative.
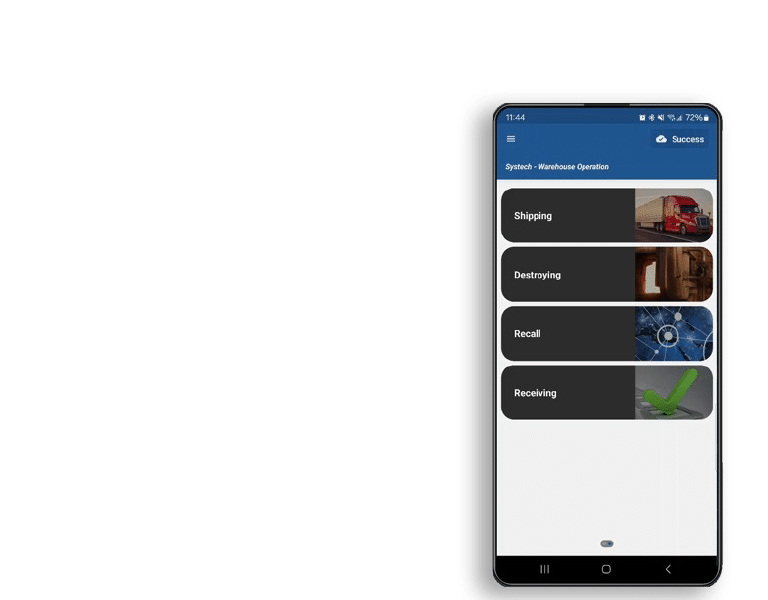
Systech offers comprehensive rework for exceptions management starting on the line and through the supply chain, with rapid deployment and seamless connectivity for everyone in the supply chain ecosystem—manufacturers, contract packagers, distributors, 3PLs, dispensaries and regulatory bodies.
What are examples of exceptions?
Systech rework is a simple plugin that offers robust features and functionality.

1. Enable connection to your L4

2. Seamlessly exchange data
3. Solve for exceptions
Systech’s solution makes exceptions handling simple, providing swift data reconciliation and resolution. Leverage the benefits of working with a trusted L1-L5 provider that can interface with any L4 platform to manage exceptions and ensure the physical matches the digital.
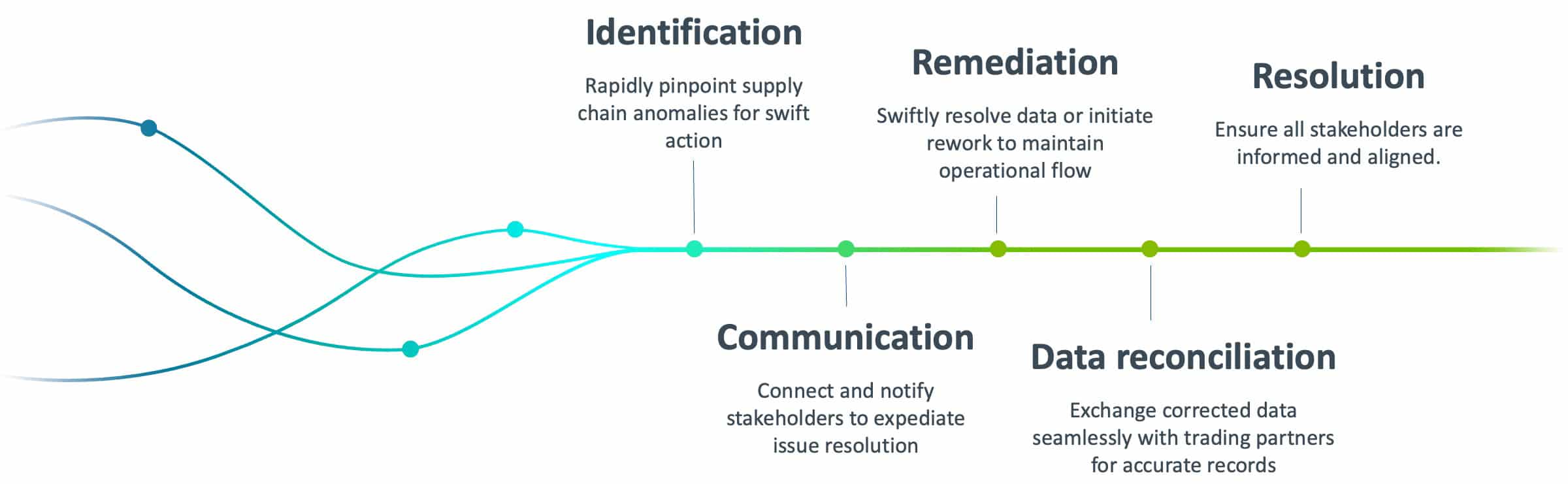
As rework pioneers, we carry 15 years of excellence—starting on the line, all the way through the pharma supply chain.



How important is aggregation to adhere to compliance and keep the pharma supply chain moving…
Misalignment exceptions – what’s the plan for the FDA declared DSCSA stabilization period ?…
Dealing with exceptions efficiently will be critical to protecting your patients health…